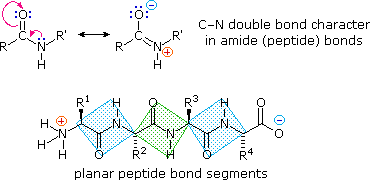
The rotation angles for the two planes are called phi and psi. The two planes can twist around the alpha carbon. The alphaĬ serves as the corner attachment point of two different planes, each which can rotate independently of the other plane. The number of conformations which a protein can adopt since these 6 atoms are forced to reside and move in a plane. In a plane, since the hybridization of the C and N are sp 2, with 120 o bond angles. These resonance structures literally force the two other atoms connected to the carbonyl C and the amide N to lie This can be accounted for by delocalizing the nonbondingĮlectron pair of the N to the carbonyl C forming a double bond, with the pi bonded electrons of the carbonyl C-O bond moving X-ray analysis shows that the the C-N bond has double bond character. The i th amino acid to the alpha amine N of the i th+1 amino acid. The peptide bonds connects the carbonyl C of For proteins, we must consider the covalent links which attach the aminoĪcids together, as well as the rotations possible in 20 different amino acids. Or dihedral angles around the methylene carbons. With fatty acid chains, we dealt only with torsion Just as saturated fatty acid chains have preferred conformations (all ttt), peptide chainsĪlso have preferred conformations. Main Chain Conformations - Cis/Trans Peptide Bonds/ Ramachandran Plots The folded (or native) shape and the unfolded (or denatured state) of proteins, in a fashion similar to how we discussed micelle The last will discuss the thermodynamics and intermolecular forces which stabilize The next section will discuss the actual processes of folding and of unfolding (denaturation), both in vitro and in vivo. The possible conformations available to proteins, just as we studied the conformations of free fatty acids and acyl chains Spontaneous process into a single unique conformation, theoretically at a global energy minimum. Proteins are covalent polymers of 20 different amino acids, which fold, to a first approximation, in a thermodynamically Note that because of processes such as the post-translational modifications to proteins we still need protein sequencing and I believe that we currently rely too heavily on DNA sequencing.In contrast to micelles and bilayers, which are composed of aggregates of single and double chain amphphiles, This is because it is now much easier to sequence DNA. Instead, since it has been worked out (mostly) how DNA codes for protein, we usually infer the protein sequence from the DNA sequence. However, it is now relatively rare to directly determine protein sequence! The very first protein sequence (bovine insulin) was determined by Fredrick Sanger in 1951-2 (note that this was more than a decade before the first nucleotide sequence). There are many different techniques for directly determining protein sequences - this wikipedia article is a decent introduction: There are also methods that have been developed to remove amino acids one at a time.īy combining theses techniques it is possible to directly determine protein sequences. This is a great question, but actually quite complicated so I'm not going to try to give a complete answer - I have given some useful links below if you wish to learn more.Įach amino acid has unique chemical properties that can be used to tell them apart.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
